Medical Device Clinical Investigation Advisory Solutions Market Size, Share, Trends, Industry Analysis and Forecast by 2033
"Latest Insights on Executive Summary Medical Device Clinical Investigation Advisory Solutions Market Share and Size
CAGR Value
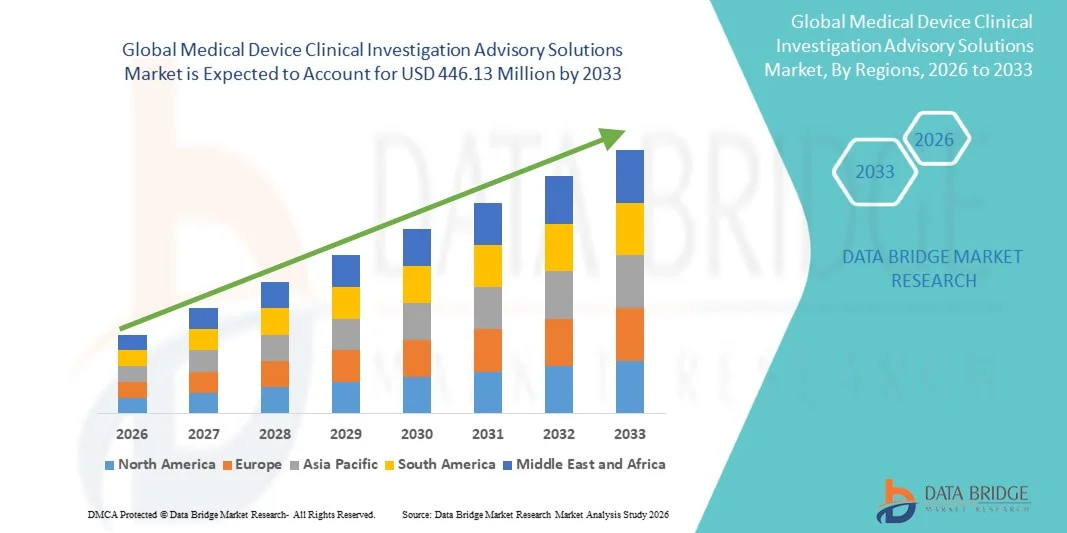
The global Medical Device Clinical Investigation Advisory Solutions market size was valued at USD 186.21 Million in 2025 and is expected to reach USD 446.13 Million by 2033, at a CAGR of 11.54% during the forecast period
This Medical Device Clinical Investigation Advisory Solutions Market research report is an absolute overview of the market that spans various aspects such as product definition, customary vendor landscape, and market segmentation based on various parameters such as type of product, its components, type of management, and geography. The report has wide-ranging and comprehensive market insights, which are based on business intelligence. The Medical Device Clinical Investigation Advisory Solutions report is generated based on the market type, size of the organization, availability on-premises, the end-users’ organization type, and the availability in areas such as North America, South America, Europe, Asia-Pacific,, and the Middle East & Africa.
This market report examines market drivers, market restraints, challenges, opportunities and key developments in the Medical Device Clinical Investigation Advisory Solutions Market What is more, this market research report also comprises of details about market analysis, market definition, market segmentation, key development areas, competitive analysis and research methodology. The Medical Device Clinical Investigation Advisory Solutions report has estimations of CAGR values which are very important for businesses in deciding upon the investment value over the time period. To gain actionable market insights to build sustainable and money-spinning business strategies with an ease, Medical Device Clinical Investigation Advisory Solutions business report is a great option.
Dive into the future of the Medical Device Clinical Investigation Advisory Solutions Market with our comprehensive analysis. Download now:
https://www.databridgemarketresearch.com/reports/global-medical-device-clinical-investigation-advisory-solutions-market
Medical Device Clinical Investigation Advisory Solutions Business Outlook
Segments
- By Device Type: The market can be segmented into in-vitro diagnostic devices, implantable devices, ophthalmology devices, surgical instruments, diagnostic imaging devices, and others. In-vitro diagnostic devices are expected to dominate the market due to the increasing demand for diagnostic tests and personalized medicine.
- By Service Type: The market can be segmented into regulatory consulting, clinical trial design, post-market surveillance, quality management, reimbursement strategy, and others. Regulatory consulting services are anticipated to hold a significant market share as companies seek guidance to navigate complex global regulatory landscapes.
- By End-User: The market can be segmented into medical device manufacturers, biotechnology companies, clinical research organizations, and others. Medical device manufacturers are expected to lead the market as they focus on enhancing the safety and efficacy of their products through clinical investigations.
Market Players
- ICON plc: A global provider of clinical research services, ICON plc offers comprehensive clinical investigation advisory solutions to medical device companies. With a strong reputation for expertise and innovation, ICON plc is a key player in the market.
- IQVIA: As a leading global provider of advanced analytics, technology solutions, and clinical research services, IQVIA offers tailored clinical investigation advisory solutions to meet the specific needs of medical device companies. Its extensive experience and global presence make it a prominent player in the market.
- Parexel International Corporation: Specializing in clinical research, consulting, and technology solutions, Parexel International Corporation provides end-to-end clinical investigation advisory services to enhance the development and commercialization of medical devices. The company’s industry knowledge and strategic approach position it as a major player in the market.
The global medical device clinical investigation advisory solutions market is experiencing robust growth driven by factors such as the increasing demand for advanced medical devices, rising focus on personalized medicine, and stringent regulatory requirements. One emerging trend in the market is the integration of digital technologies such as AI and IoT to enhance clinical investigation processes and improve patient outcomes. This trend is expected to drive market growth as companies look to leverage technology for more efficient and effective clinical investigations.
Another key driver of market growth is the growing emphasis on post-market surveillance and quality management services. With regulatory bodies placing greater scrutiny on the safety and effectiveness of medical devices throughout their lifecycle, companies are increasingly investing in post-market surveillance and quality management solutions to ensure compliance and mitigate risks. This shift towards proactive risk management is creating opportunities for service providers offering comprehensive advisory solutions tailored to the unique needs of medical device companies.
Moreover, the market is witnessing increasing collaboration between medical device manufacturers, clinical research organizations, and regulatory consulting firms to streamline the clinical investigation process and expedite market approval. This collaborative approach is driven by the need for faster time-to-market and the growing complexity of regulatory requirements, prompting industry players to work together to navigate challenges and accelerate product development.
Furthermore, market players are focusing on expanding their service portfolios and geographic presence to cater to the growing demand for clinical investigation advisory solutions globally. This strategy enables companies to better serve their clients and stay ahead of competitors in a rapidly evolving market landscape. Additionally, the adoption of innovative business models such as outcome-based pricing and risk-sharing agreements is becoming more prevalent in the market, allowing for more flexible and cost-effective engagement between service providers and medical device companies.
Overall, the global medical device clinical investigation advisory solutions market presents significant growth opportunities for companies offering specialized services in regulatory consulting, clinical trial design, post-market surveillance, and quality management. With the increasing complexity of regulatory requirements and the continuous evolution of medical technologies, the demand for expert advisory solutions is expected to remain strong, driving market expansion and innovation in the years to come.The global medical device clinical investigation advisory solutions market is poised for substantial growth as the industry continues to evolve in response to increasing demand for advanced medical technologies and personalized medicine. One of the key trends shaping the market is the integration of digital technologies like artificial intelligence (AI) and the Internet of Things (IoT) into clinical investigation processes. This integration not only enhances efficiency in conducting investigations but also improves patient outcomes by leveraging data-driven insights for decision-making. As companies embrace these digital advancements, we can expect a significant transformation in how clinical investigations are conducted, leading to better outcomes for both patients and healthcare providers.
Moreover, the emphasis on post-market surveillance and quality management services is driving market growth as regulatory bodies tighten their scrutiny on medical device safety and effectiveness. Companies are recognizing the importance of proactive risk management and compliance with regulations throughout the product lifecycle, leading to increased investment in post-market surveillance and quality management solutions. Service providers offering comprehensive advisory services tailored to meet these evolving needs are well-positioned to capitalize on this growing demand and drive innovation in the market.
Another important driver of market growth is the trend towards collaboration among key industry stakeholders, including medical device manufacturers, clinical research organizations, and regulatory consulting firms. By working together to streamline the clinical investigation process and navigate regulatory complexities, these players are able to accelerate product development and expedite market approval. This collaborative approach not only enhances efficiency but also fosters innovation in the industry, paving the way for more effective and timely delivery of cutting-edge medical devices to market.
Furthermore, market players are increasingly focusing on expanding their service offerings and global footprint to cater to the rising demand for clinical investigation advisory solutions worldwide. By diversifying their portfolios and adopting innovative business models such as outcome-based pricing and risk-sharing agreements, service providers can better meet the needs of medical device companies and stay competitive in a rapidly evolving market landscape. This strategic approach allows companies to differentiate themselves, drive growth, and foster long-term relationships with clients in an increasingly dynamic and competitive market.
In conclusion, the global medical device clinical investigation advisory solutions market is experiencing significant growth driven by technological advancements, regulatory requirements, and collaborative partnerships among industry stakeholders. As companies continue to prioritize patient safety, regulatory compliance, and innovation in medical device development, the demand for specialized advisory services is expected to remain strong, creating ample opportunities for market expansion and differentiation. By staying abreast of market trends, embracing digital technologies, and fostering strategic collaborations, players in this space can position themselves for success and contribute to the advancement of healthcare technologies globally.
Analyze detailed figures on the company’s market share
https://www.databridgemarketresearch.com/reports/global-medical-device-clinical-investigation-advisory-solutions-market/companies
Medical Device Clinical Investigation Advisory Solutions Market – Analyst-Ready Question Batches
- What is the current demand volume of the Medical Device Clinical Investigation Advisory Solutions Market?
- How is the market for Medical Device Clinical Investigation Advisory Solutions expected to evolve in the next decade?
- What segmentation criteria are applied in the Medical Device Clinical Investigation Advisory Solutions Market study?
- Which players have the highest market share in the Medical Device Clinical Investigation Advisory Solutions Market?
- What regions are assessed in the country-level analysisfor Medical Device Clinical Investigation Advisory Solutions Market?
- Who are the top-performing companies in the Medical Device Clinical Investigation Advisory Solutions Market?
Browse More Reports:
Global Cell Therapy Market
Global Electronic Specialty Gas Market
Global Minimally Invasive Surgery Market
Global Digital Twin Market
Global Isocyanate Market
Global Used Cooking Oil Market
Global Hair Loss Treatment Products Market
Global Vegan Dog Food Market
Global Agricultural Biotechnology Market
Global Epigenetics Diagnostic Market
Global Mobility as A Service Market
Global Nurse Call System Market
Global Pine Nuts Market
Global Revenue Cycle Management (RCM) Market
Global Shea Butter Market
About Data Bridge Market Research:
An absolute way to forecast what the future holds is to comprehend the trend today!
Data Bridge Market Research set forth itself as an unconventional and neoteric market research and consulting firm with an unparalleled level of resilience and integrated approaches. We are determined to unearth the best market opportunities and foster efficient information for your business to thrive in the market. Data Bridge endeavors to provide appropriate solutions to the complex business challenges and initiates an effortless decision-making process. Data Bridge is an aftermath of sheer wisdom and experience which was formulated and framed in the year 2015 in Pune.
Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC : +653 1251 975
Email:- corporatesales@databridgemarketresearch.com
"
- الاقتصاد والتجارة
- فن
- كورسات
- الحرف اليدوية
- الطعام والشراب
- الألعاب والترفيه
- الصحة
- تكنولوجيا
- أخرى
- دين
- رياضة